BOXED WARNING: HEPATOCELLULAR INJURYAQVESME can cause serious hepatocellular injury. Measure liver laboratory tests (ALT, AST, alkaline phosphatase and total bilirubin with fractionation) at baseline and every 4 weeks for 24 weeks and then as clinically indicated. Avoid use of AQVESME in patients with cirrhosis. Discontinue AQVESME if hepatic injury is suspected.
Because of the risk of hepatocellular injury, AQVESME is available only through a restricted program under a Risk Evaluation and Mitigation Strategy (REMS) called the AQVESME REMS.
WARNINGS AND PRECAUTIONS
Hepatocellular Injury
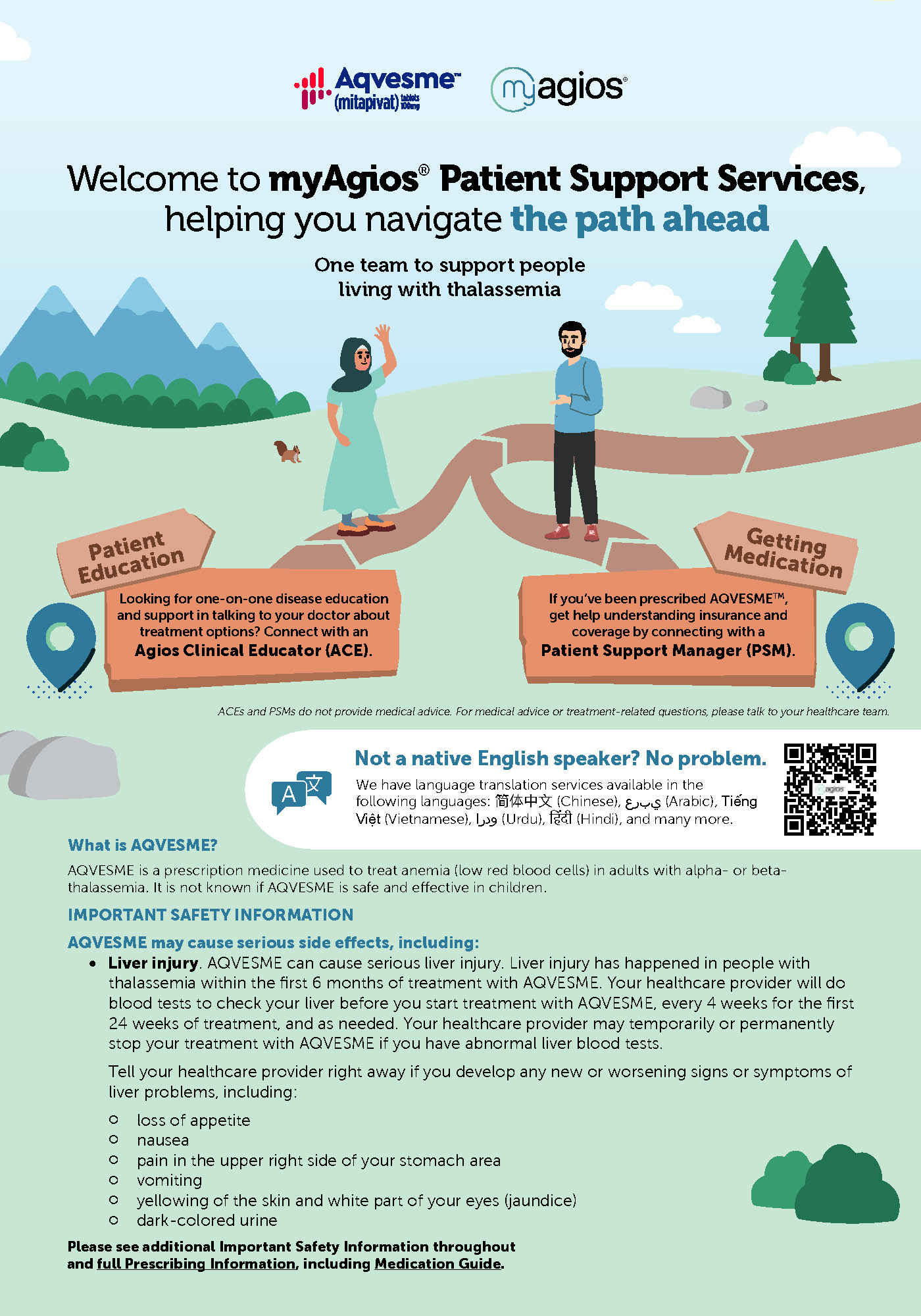
AQVESME can cause hepatocellular injury. Avoid use of AQVESME in patients with cirrhosis. In patients with thalassemia treated with AQVESME, liver injury with and without jaundice has been observed within the first 6 months of exposure. Obtain liver tests (including ALT, AST, alkaline phosphatase, total bilirubin with fractionation) prior to the initiation of AQVESME, then every 4 weeks for the first 24 weeks, and as clinically indicated thereafter. Interrupt AQVESME if clinically significant increases in liver tests are observed or alanine aminotransferase is >5 times the upper limit of normal (ULN). Complete a comprehensive evaluation to rule out other causes of liver injury when drug-induced liver injury is suspected. Discontinue AQVESME if hepatocellular injury due to AQVESME is suspected.
Symptoms and signs of early liver injury may mimic those of thalassemia. Advise patients to report new or worsening symptoms of loss of appetite, nausea, right-upper-quadrant abdominal pain, vomiting, scleral icterus, jaundice, or dark urine while on AQVESME treatment.
During the double-blind period, 2 of 301 patients (0.66%) with thalassemia treated with AQVESME experienced adverse reactions suggestive of hepatocellular injury. Three additional patients experienced adverse reactions suggestive of hepatocellular injury during the open-label extension periods after switching from placebo to AQVESME. Of these 5 patients, 2 had serious liver injury requiring hospitalization, including 1 patient who developed jaundice (peak bilirubin 32 mg/dL). Another patient developed jaundice (peak bilirubin 4 mg/dL) without requiring hospitalization. These reactions were characterized by a time to onset within the first 6 months of treatment with peak elevations of alanine aminotransferase of >5×ULN with or without jaundice. All patients discontinued treatment with AQVESME, and these reactions improved upon treatment discontinuation.
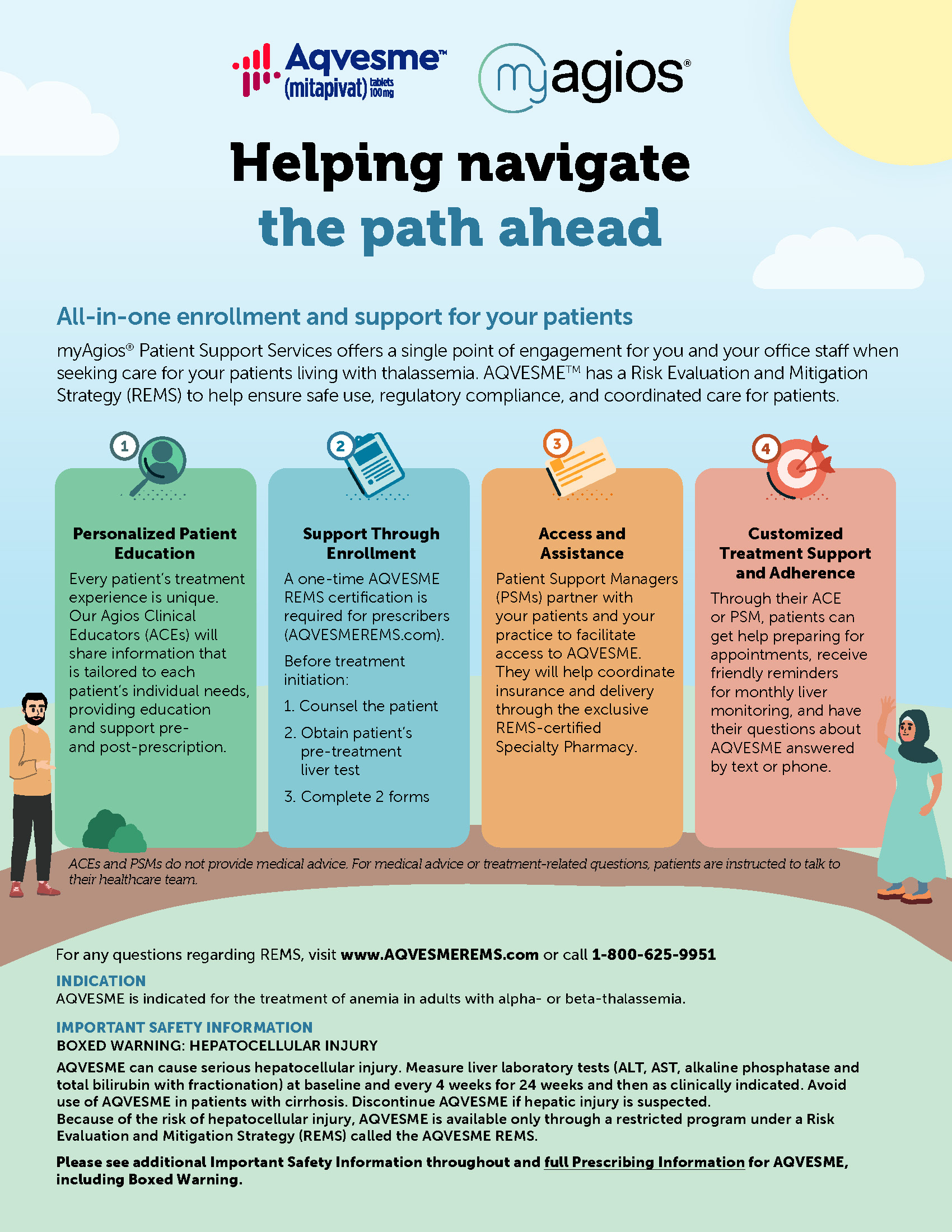
AQVESME REMS
AQVESME is available only through a restricted program under a REMS called the AQVESME REMS because of the risk of hepatocellular injury.
ADVERSE REACTIONS
The most common adverse reactions among patients taking AQVESME were headache and insomnia.
DRUG INTERACTIONS
- Strong CYP3A Inhibitors and Inducers: Avoid concomitant use.
- Moderate CYP3A Inhibitors: Avoid concomitant use.
- Moderate CYP3A Inducers: Consider alternatives that are not moderate inducers. If there are no alternatives, see full Prescribing Information for recommended dosage for drug interactions with moderate CYP3A inducers.
- Sensitive CYP3A Substrates, including hormonal contraceptives: Avoid concomitant use with substrates that have narrow therapeutic index.
- CYP2B6, CYP2C, and UGT1A1 Substrates: Monitor patients for efficacy of the substrates with narrow therapeutic index.
- P-gp Substrates: Monitor patients for adverse reactions of the substrates with narrow therapeutic index.
HEPATIC IMPAIRMENT
Avoid use of AQVESME in patients with cirrhosis (Child-Pugh Class A, B, or C).
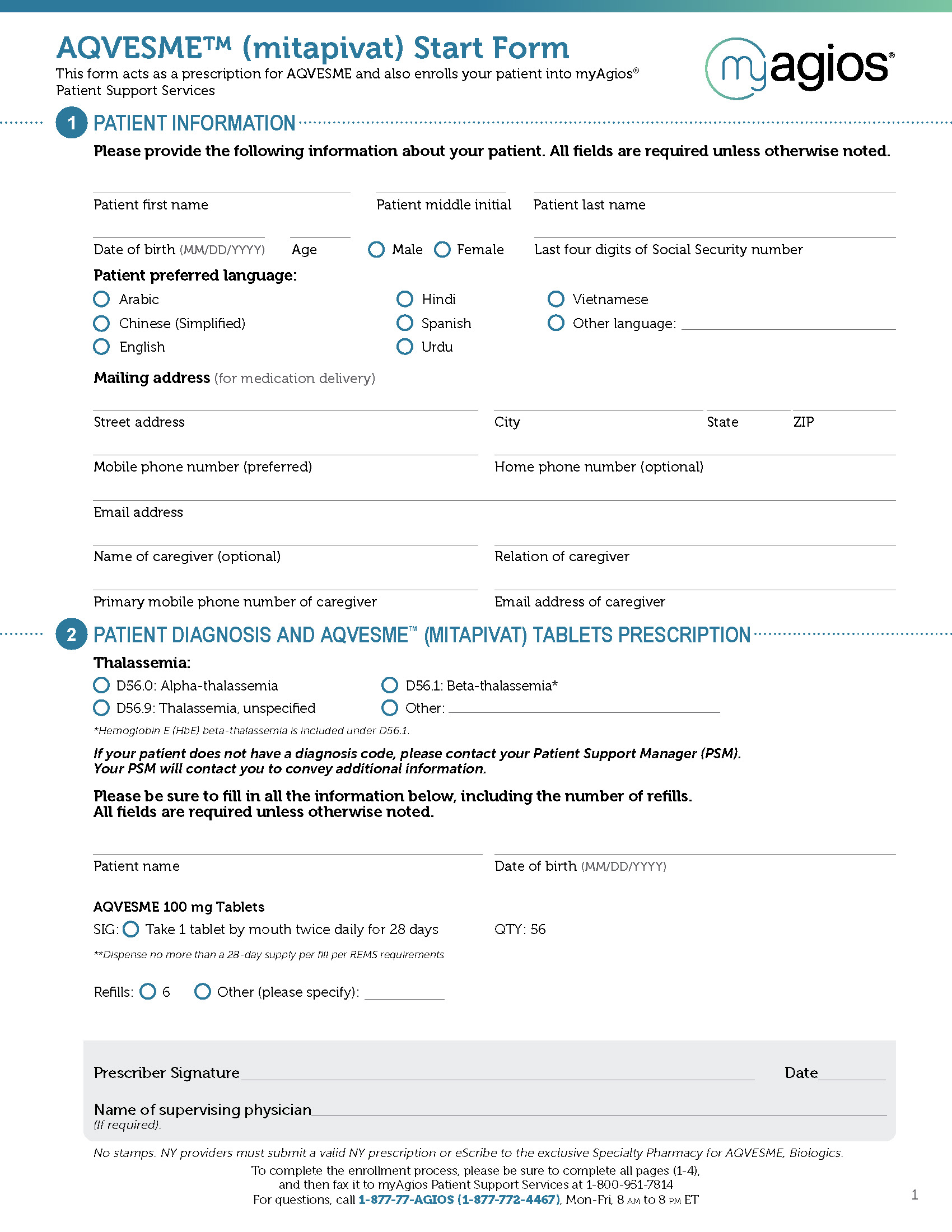
INDICATION
AQVESME is indicated for the treatment of anemia in adults with alpha- or beta-thalassemia.
Please see full Prescribing Information for AQVESME, including Boxed Warning.